An industrial plant was required to reduce its steam production due to commercial and market-driven decisions. After inspecting both boilers, it was recommended that the existing layup procedure be modified, as it was not the most technically appropriate method for the situation.
The plant elected to operate one boiler continuously for six months while placing the second unit in hibernation, then alternate the roles for the remaining six months of the year. The selected preservation technique was the so‑called cascade hibernation or cascade layup method. In this approach, the continuous blowdown from the operating boiler is routed to the bottom of the idle boiler until the entire volume is filled, after which the water is discharged through the continuous blowdown line.
Figure 1 illustrates this layup option.
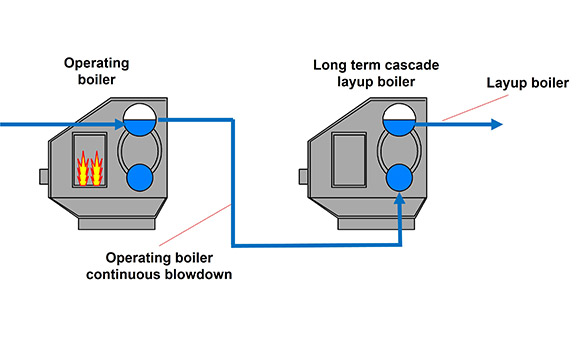
Figure 1 – Cascade boiler lay-up procedure.
In situations where layup is required for many months and years, this method is not suitable due to the low residual oxygen scavenger/passivating agent in the normal operation of the operating boiler. In long layups, high residual oxygen scavenger is always recommended, with the essential objective of protection against dissolved oxygen corrosion. Unfortunately, in the first few years, the plant team had a false impression from visual inspections that the procedure was working perfectly well. However, this is a known phenomenon: when a protective oxide layer (such as magnetite) exists, in the face of an adverse corrosive situation, there is a latent period before this protective layer is affected. After the protective layer is compromised, vigorous corrosion becomes evident. Figure 2 is a typical graphic representation of different corrosion velocities and loss of mass according to specific conditions. As described by Vicente Gentil (Corrosion, 5th edition, page 110):
Curve A - Occurs when the metal surface does not change, the corrosion product is inert, and the concentration of the corrosive agent is constant.
Curve B - Similar to A, except that there is an induction period which is related to the time spent by the corrosive agent to destroy previously existing protective films.
Curve C - Corrosion rate is inversely proportional to the amount of corrosion product formed; the corrosion product is insoluble and adheres to the surface.
Curve D - Corrosion velocity increases rapidly, corrosion products are soluble, and anodic areas increase.
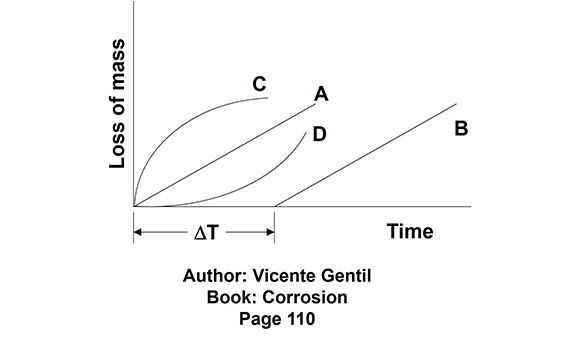
Figure 2 - Typical corrosion velocities.
After several years of using this approach, an inspection revealed severe and widespread corrosion in both boilers. Significant and unusually localized areas of corrosion caused by dissolved oxygen were identified. Unfortunately, this damage resulted from selecting an inappropriate layup procedure. Over the long term, other well‑established layup methods provide far more reliable protection for critical assets such as high‑pressure boilers.
Figure 3 shows the undesirable results observed during a boiler inspection.

Figure 3 - Severe corrosion on boiler internal surfaces.
Effective water treatment relies on understanding each operation’s unique characteristics and continuously optimizing every step of the process. At Solenis, we combine innovative chemistries, advanced monitoring and control systems, and expert support to help our partners overcome these challenges and operate more efficiently and sustainably.
Learn more about how our water treatment solutions can help you achieve your operational and sustainability goals: https://www.solenis.com/en/products-and-services/water-treatment/
Reference: Corrosão (Corrosion), author Vicente Gentil.